Describe Some of the Chemical Effects of Acids and Bases
Is obviously an acid. Acids Bases and the pH Scale.
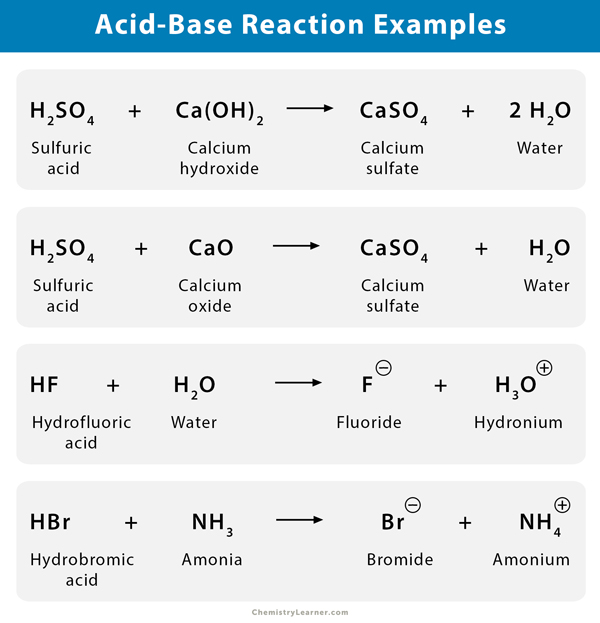
Acid Base Reaction Definition Examples And Uses
Acids bases and pH.
. Acid burns usually cause immediate pain and tissue damage. Bases turn red litmus to blue 2. Some weak acids and bases are used in foods for example vinegar is a base and citric acid From lemons etc.
Citric acid found in oranges and lemons is one example in which the sour taste is related to the fact that the chemical is an acid. Acidic things taste sour. Frequently react with metals to form H 2.
Sour vinegar Bitter baking soda Smell. Lactic acid-acid found in yogurt butter and milk. Where are acids and basis commonly used.
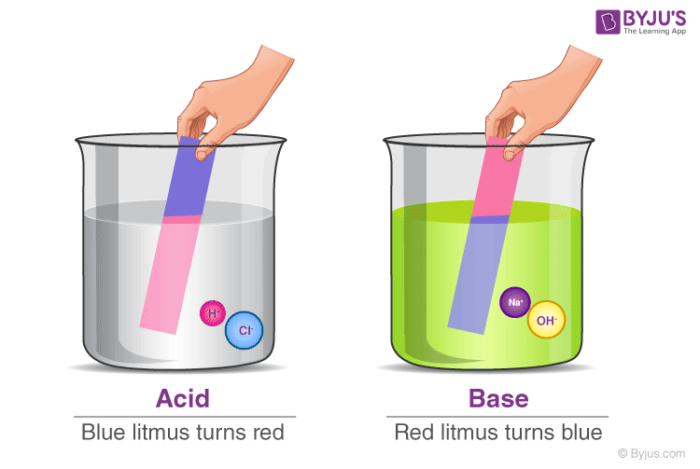
Acids turn methyl orange to red Bases effect indicators. Determine whether a solution is acid basic or neutral. Concentrated acids and bases are corrosive and cause chemical burns if they come into contact with the skin eyes or internal organs.
Ranging in pH scale from 0 to 7. An acid is a proton donorBecause a hydrogen atom without its electron is a proton any substance that releases hydrogen ions H. Molecules that are bases usually have a bitter taste like caffeine.
Water which has both acid and base is considered chemically neutral having a pH of 7. It would have a bitter taste. Normally systemic acid-base balance is well regulated with arterial pH between 736 and 744.
Would turn litmus paper blue. Base has a characteristic to neutralize acid. Our mission is to provide a free world-class education to anyone anywhere.
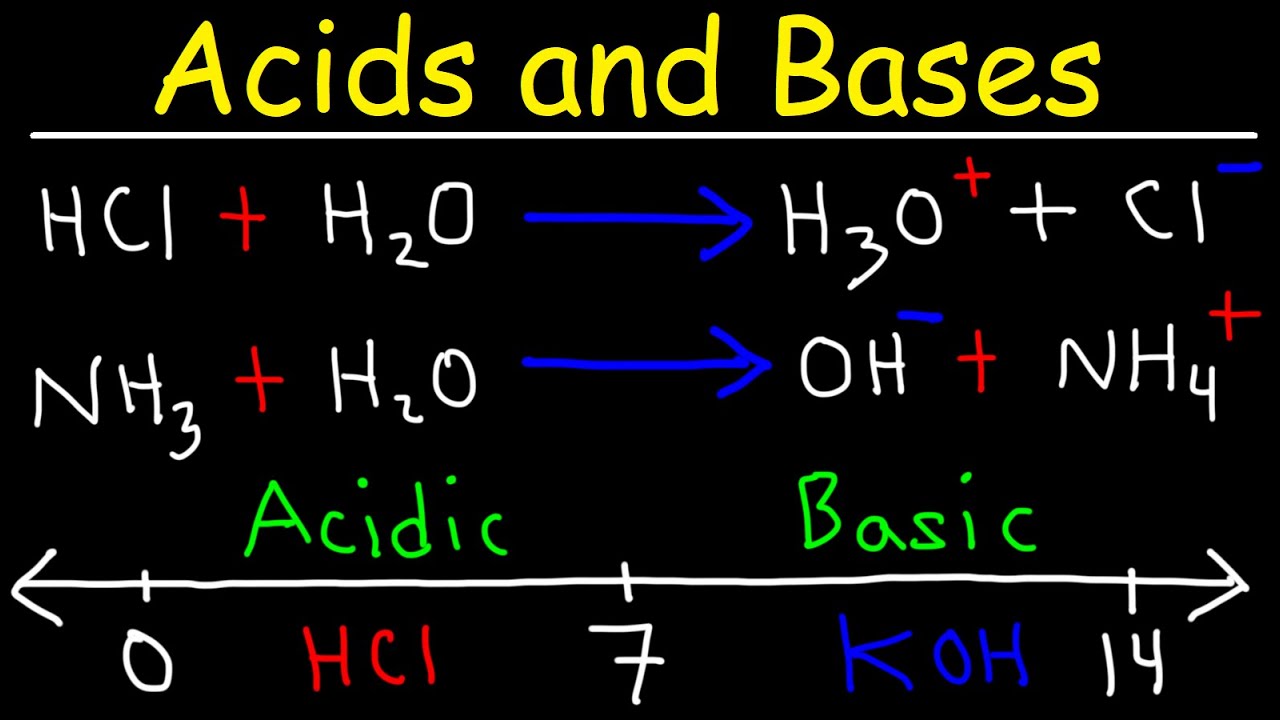
The Bronsted-Lowry theory defines an acid as a proton donor and a base as a proton acceptor. Bases turn phenolphthalein to purple Acids neutralize bases producing a salt and water. The Arrhenius theory of acids and bases states that an acid generates H ions in a solution whereas a base produces an OH ion in its solution.
It can conduct electrical current. Acids are common chemicals and can be found everywhere even in our food. Acids would release hydrogen ions h when mixed with water.
Acids and bases can help neutralize each. It helps in the breakdown of all. Bases would feel slippery and solid in nature except for ammonia which is gaseous.
Other acids are weak electrolytes that exist primarily in a non-ionized form when dissolved in water. Explain the importance of buffers in organisms. Lemons vinegar and sour candies all contain acids.
Acids Bases Taste sour Taste Bitter pH less than 7 pH greater than 7Examples of acids. It transforms red litmus paper into blue. Can eat through some metals.
When the metal iron disappears in an acid solution we can describe the acid as being what. What are the properties of acids and bases. Bases would release hydroxide ionsOH- when mixed with water.
Some dishes just need some acid eg. Would turn litmus paper red. Now the water molecules give up their own protons to the base and become what are known as hydroxide Hy-DROX-ide molecules.
Basic or alkaline things taste soapy. To gauge whether something is an acid or base and how strong it is chemists employ the pH scale. Intracellular pH is usually approximately 72.
Science and technology makes good use of acids and bases. Examples of Acids and Bases in Everyday Life. They are also used in many household cleaning products baking soda and to make fertilizer for crops.
They are the chemical substances. Acids bases and pH. Acids and bases can be defined via three different theories.
What is released when an acid is dissolved in water. Hydrochloric Acid HCl Hydrochloric acid is the strong acid which is found inside our body in the gastric juice. Some common acids are hydrochloric acid nitric acid carbonic acid and acetic acid.
Biology is brought to you with support from the Amgen Foundation. Lemon juice to work well. In contrast you may not notice right away if your skin contacts a base.
However mild acids and bases are common and relatively harmless to us. Usually no smell except NH 3 Texture. Acids change the color of certain acid-base indicators.
It keeps blue litmus paper blue. Bases turn methy l orange to yellow 3. Mix water with a base and that water will play the part of the acid.
Lets check its everyday uses. Car batteries use a strong acid called sulphuric acid. Please respond if this did not fully help you understand acids.
The body has many molecules and compounds called acids and bases that can alter body functions. For instance chronic metabolic acidosis can be associated with decreased bone density nephrolithiasis muscle wasting and progression of CKD 13. Of course acid isnt just about chemical reactions.
A base is a substance with a pH value greater than 7. Strong acids are corrosive and strong bases are caustic. Some acids are strong electrolytes because they ionize completely in water yielding a great many ions.
The name acid gives us sensory images of Sourness. Acids bases and pH. React with many oils and fats.
It has a bitter taste. The terms acid and base describe chemical characteristics of many substances that we use daily. Turns litmus paper red.
How acidic or alkaline a substance is the pH of the substance can be measured using the pH scale a continuous range that stretches from below 0 to above 14. Hydrofluoric acid penetrates the skin rapidly and may even result in bone damage. Acids and bases have some general properties.
Acids and bases The pH scale. Bases are termed alkaline meaning that they neutralize acids. Describe the pH scale and its relationship to acidic and basic solutions.
Some can burn skin. Acids The ionization of a group of acids in a given solvent may be used as a measure of acid strength and in such cases the solvent serves as a common reference base. Biology is brought to you with support from the.
7 rows Tertiary amines and bulky secondary amines are poorer nucleophiles and behave exclusively as bases. Bases make solutions that are slippery. Acids have a sour taste.
Many acids have a sour taste. Acids turn blue litmus to red 2. It is also a lot about flavour and balance of a dish.
This is caused by acidbase reactions with the molecules that give the red cabbage its colour. An acid is a solution with a value between 1 and 699 on the pH scale a chemical gauge of acidity and alkalinity that goes from 1 to 14. It has a pH level of more than 7.
4 properties of acids. Both can cause severe skin damage that feels like a burn. Chemical reactions between the acid and lead plates in the battery help make electricity to start the car.

Acids And Bases Chapter 14 Acid Base Chemistry Acid Base Chemistry Characteristics Acidsbases Ph Less Than 7ph Greater Than 7 Donates H Ionsaccepts Ppt Download
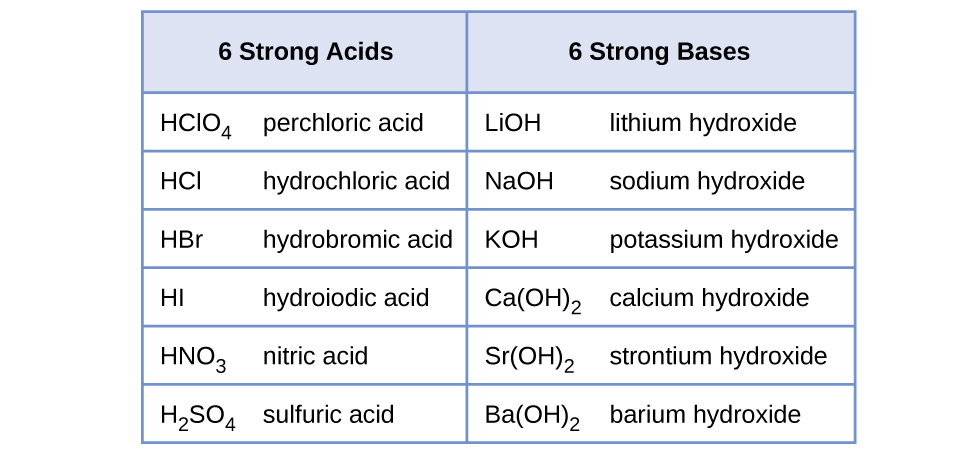
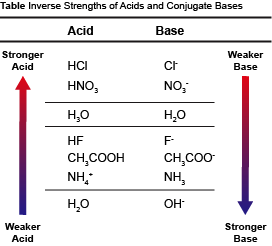
14 3 Relative Strengths Of Acids And Bases Chemistry
Difference Between Acid And Base Differences Btw Acid Base In Tabular Form
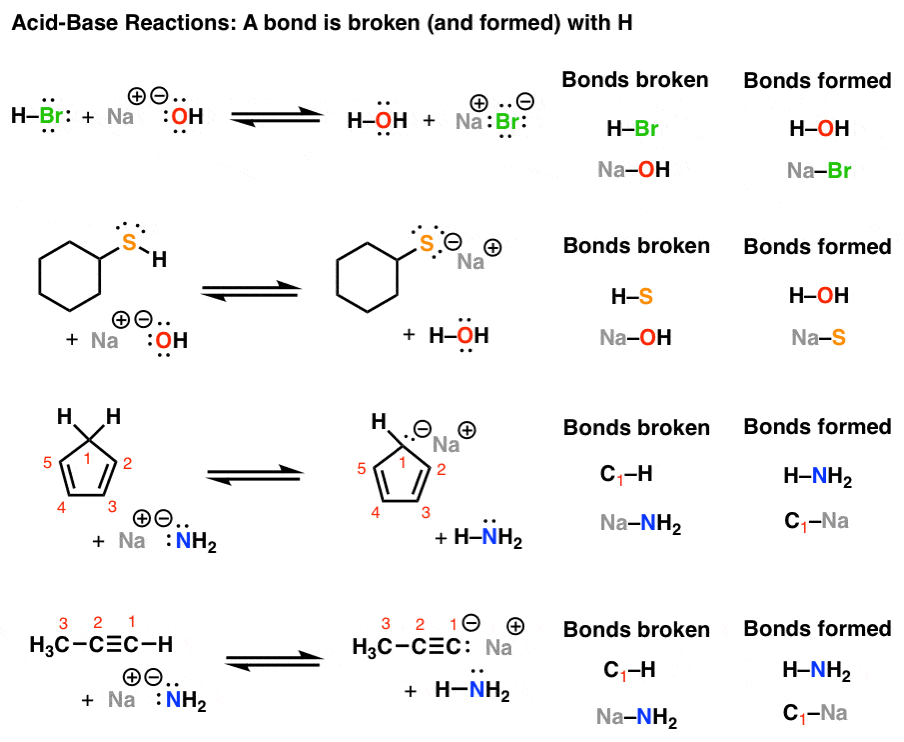
Introduction To Acid Base Reactions Master Organic Chemistry
Acids And Bases What Are Acids And Bases Shmoop

Organic Acids And Bases Chemistry Steps
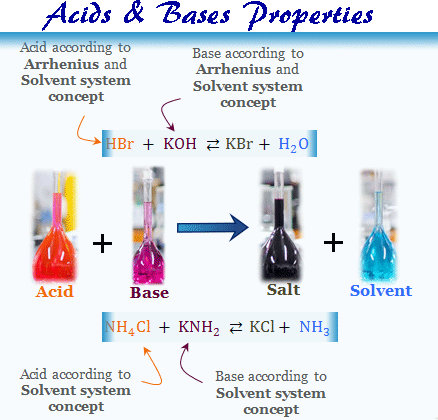
Acids Bases Properties Definition Concept Theory Examples

Illustration Chemistry Acid Base Reaction Chemical Stock Vector Royalty Free 1486011773

Properties Of Acids And Bases Physical And Chemical Properties With Examples
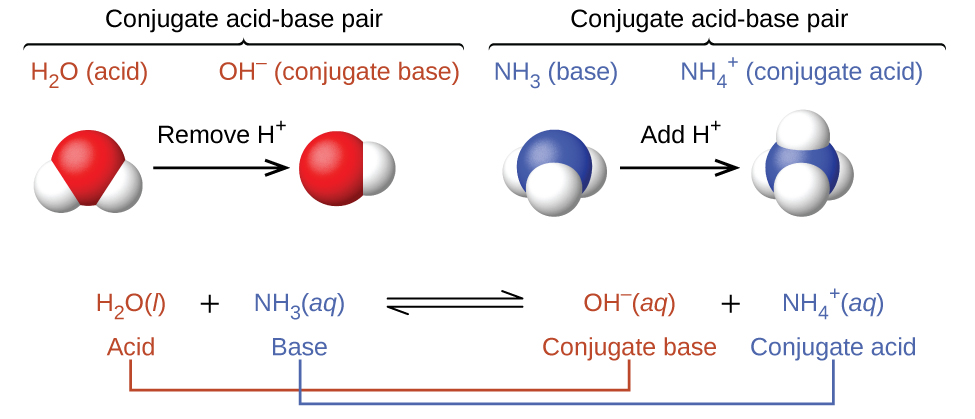
14 1 Bronsted Lowry Acids And Bases Chemistry

Acids And Bases Basic Introduction Chemistry Youtube
Acid Base Chemistry Chemistry Encyclopedia Reaction Water Metal Gas Number Equation Salt Property

Relative Strengths Of Acids And Bases Chemistry For Majors
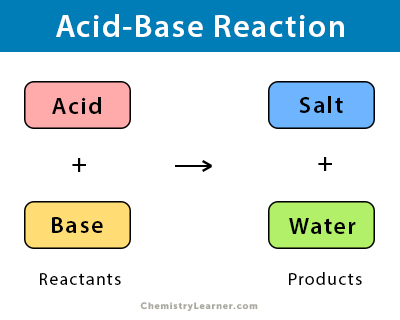
Acid Base Reaction Definition Examples And Uses

Acids And Bases What Are Acids And Bases Shmoop

Chemistry Acid Base Reaction Chemical Equations 13 Of 38 Types Of Reactions Vii Youtube
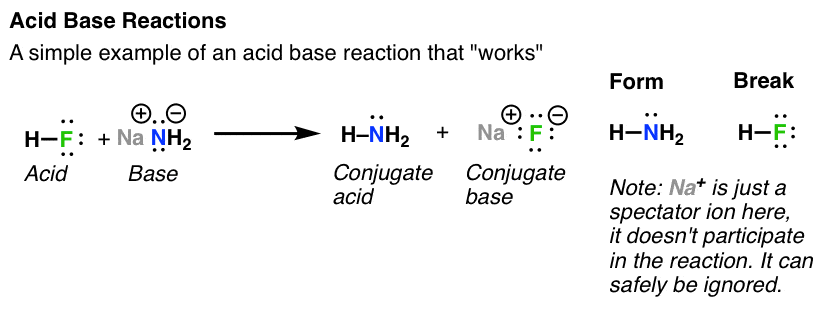
Acid Base Reactions In Organic Chemistry Master Organic Chemistry

Comments
Post a Comment